Antihypertensive Combinations – What do the Guidelines Tell Us?
Hypertension affects more than eight million individuals in the UK. Over 1.2 million of the patients currently being treated have neither reached Quality and Outcomes Framework targets (150/90 mmHg) nor clinical standards (140/90 mmHg), and approximately two-thirds of diagnosed patients are prescribed two or more antihypertensive drugs.1 Poor patient adherence is a contributing factor to the current high incidence of uncontrolled high blood pressure (BP). In fact, half of hypertensive patients do not adhere to the prescribed regimens and fail to persist with treatment after one year,2,3 increasing their risk for stroke, myocardial infarction and kidney failure. Although the underlying causes of low adherence rates are varied and complex, with pill burden, side effects and asymptomatic hypertension as the main culprits, possible solutions for improving compliance must involve patients, healthcare providers, clinical institutions and the community.4
Current guidelines for hypertension treatment recommend early combination therapy for patients with stage II hypertension or at high cardiovascular (CV) risk. The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC7) considers the use of two drugs when systolic blood pressure (SBP) is more than 20 mmHg above goal and diastolic blood pressure (DBP) is 10 mmHg higher than the ideal target.5 The joint statement of the European Society of Hypertension and the European Society of Cardiology (2007 ESH-ESC) also recognises that a combination of two drugs may offer advantages for new and advanced patients, particularly in the presence of a high CV risk warranting early BP control.6 More recently, the ESH has reappraised these guidelines in view of newly gathered data on the effectiveness of combination therapy versus other therapeutic approaches, namely dose escalation and monotherapy,7 and concluded that an effective BP control can only be achieved in most patients by combining antihypertensive drugs of distinct classes.6 Such combination therapy can also benefit newly diagnosed high-risk patients and contribute to higher compliance rates when provided as fixed-dose combinations.8
The Action in Diabetes and Vascular disease: preterAx and diamicroN modified-release Controlled Evaluation (ADVANCE)9 trial and the Perindopril pROtection aGainst REcurrent Stroke Study (PROGRESS),10 as well as the Hypertension in the Very Elderly Trial (HyVET),11 provide evidence for superior clinical outcomes with an angiotensin-converting enzyme inhibitor (ACEI) combined with a diuretic. In addition, the Anglo-Scandinavian Cardiac Outcomes Trial – Blood Pressure Lowering Arm (ASCOT-BPLA)12 and the Avoiding Cardiovascular events through COMbination therapy in Patients LIving with Systolic Hypertension (ACCOMPLISH)13 study showed effective BP control with an ACEI and a calcium channel blocker (CCB). An angiotensin II receptor blocker (ARB)/diuretic combination also yielded promising results in the Losartan Intervention For Endpoint reduction (LIFE) study,14 but there are currently no data to support the use of an ARB/CCB combination.
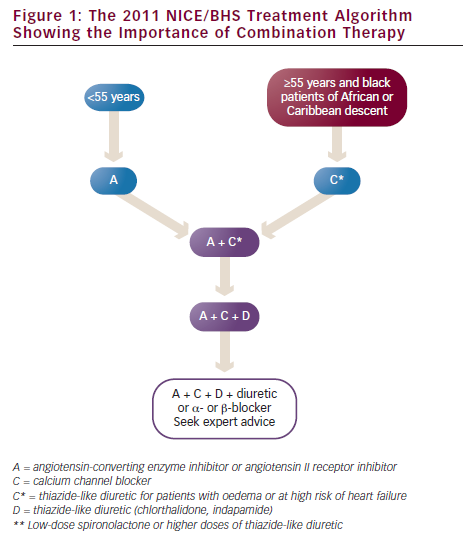
In ASCOT-BPLA, amlodipine/perindopril performed better than atenolol/thiazide on secondary endpoints of all-cause mortality (738 versus 820 events; p=0.0247), CV mortality (263 versus 342 events; p=0.0010), stroke (327 versus 422; p=0.0003) and non-fatal myocardial infarction (MI) (excluding silent MI) plus coronary heart disease (CHD) (390 versus 444; p=0.0458) in patients at high risk.12 Data from this study were partly responsible for the formulation, in 2006, of specific guidelines by the National Institute for Health and Clinical Excellence (NICE), in partnership with the British Hypertension Society (BHS), recommending the use of an ACEI combined with a CCB or, as an alternative, with a thiazide-type diuretic as second-line therapy. The 2006 NICE/BHS guidelines also recommended, for first-line therapy, monotherapy with an ACEI in patients younger than 55 years, and a CCB or thiazide-type diuretic (e.g. chlorthalidone, indapamide) in older or black patients; for third-line therapy, triple therapy with an ACEI, CCB and a diuretic was recommended, adding later another diuretic and an α- or b-blocker, if necessary.15
This treatment algorithm was later revised in order to incorporate new evidence from the ACCOMPLISH study, which clearly showed the superiority of an ACEI/CCB (benazepril/amlodipine) versus an ACEI/diuretic (benazepril/hydrochlorothiazide) combination in reducing CV events and CV-related death (relative risk reduction [RRR] of 20 %, hazard ratio [HR]=0.80, p<0.001; and 20 %, HR=0.80, p=0.08, respectively).13 Therefore, the 2011 NICE/BHS guidelines recommend an ACEI or ARB for first-line hypertension therapy in patients younger than 55 years, and a CCB in older or black patients (monotherapy with a diuretic is no longer recommended); for second-line therapy a combination of ACEI (or ARB) and a CCB is recommended adding later a diuretic and an α- or b-blocker, if necessary (see Figure 1). Although relying on different levels of evidence, the current guidelines recognise the need for combination therapy to keep hypertensive patients on target and favour an ACEI/CCB combination for the majority of patients.
Angiotensin-converting Enzyme Inhibition, the Sine Qua Non of Combination Therapy
Although recommended and widely used in the treatment of CV and renal disease, ACEIs and ARBs have distinct effects on the renin–angiotensin system. ACEIs reduce the synthesis of angiotensin II and increase nitric oxide (NO) production via bradykinin; by blocking angiotensin II type 1 receptors, ARBs increase angiotensin II levels and stimulate angiotensin II type 2 receptors, which have potential harmful effects, but they do not affect bradykinin or NO.16 Such overstimulation of AT2R by ARBs may increase levels of matrix metalloproteinase-1, activate leukocytes and inhibit the growth of vascular smooth muscle cells leading to plaque destabilisation and rupture.17
Bradykinin, a promising therapeutic target in the control of hypertension,18 appears to play an essential role in cardiac volume maintenance and presents antioxidant, anti-remodelling and antithrombotic properties. The potent stimulatory action it exerts on tissue plasminogen activator induces vasodilation via release of prostacyclin and NO, which in turn contributes to oxidative stress reduction.19–22
ACEIs show significant survival benefits and prevent the occurrence of MI in high-risk populations (including patients presenting with heart failure and a history of MI and diabetes), suggesting that these two classes may not be interchangeable.23 In particular, ACE inhibition with perindopril reduces risk of MI (RRR=24 %, p<0.01) in patients with prior coronary heart disease and/or revascularisation, as shown in the EUropean trial of Reduction Of cardiac events with Perindopril in stable coronary Artery disease (EUROPA) study.24 Also, the Blood Pressure Lowering Treatment Trialists' Collaboration (BPLTTC) calculated an estimated CHD odds ratio (OR) at zero blood pressure reduction (i.e. considering only BP-lowering independent effects) of 0.91 (95 % confidence interval [CI] 0.86–0.97) for ACEIs and 1.08 (CI 0.83–1.39) for ARBs (p=0.003), based on the results of 26 large-scale comparative trials.25 Moreover, a meta-analysis and sequential analysis of data from over 100,000 patients participating in CV randomised trials showed no beneficial effect for the outcome of MI and CV mortality with ARBs, despite successful BP lowering.26 Finally, a recent meta-regression analysis showed that ACEIs reduced all-cause mortality by 10 % compared with 0 % for ARBs (overall HR=0.90, 95 % CI 0.84-0.97, p=0.004 for ACEIs versus HR=0.99, CI 0.94–1.04, p=0.683 for ARBs).27
These results supported the information previously provided by ASCOT, which showed superior survival benefits and SBP (mean difference=2.7 mmHg) and DBP (mean difference=1.9 mmHg) reductions for the ACEI/CCB combination versus a b-blocker/diuretic combination, with much higher reductions in coronary events and stroke than predicted based on the number of events due to BP reductions alone (14 % versus 4–8 % and 23 % versus 11–14 %, respectively).12
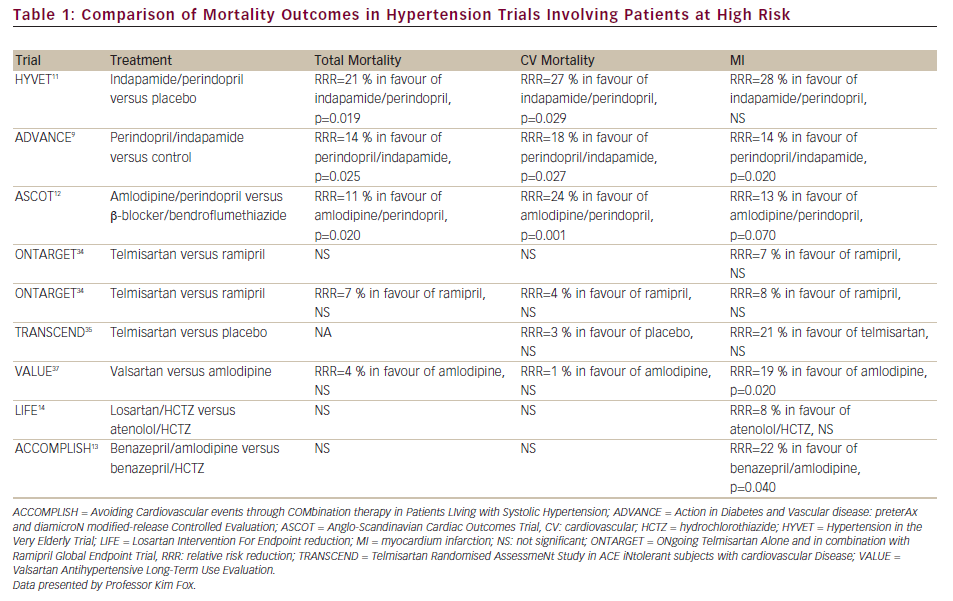
Remarkably, only three studies – ASCOT (amlodipine/perindopril versus b-blocker/bendroflumethiazide),12 ADVANCE (perindopril/indapamide versus placebo)9 and HyVET (indapamide/perindopril versus placebo)11 - showed a reduction in all-cause and CV mortality for ACEIs. For HyVET, these reductions were 21 % (p=0.019) and 27 % (p=0.029); for ADVANCE, 14 % (p=0.025) and 18 % (p=0.027); and for ASCOT, 11 % (p=0.02) and 24 % (p=0.001), respectively. However, ARBs increased frequency of CV events despite their BP-lowering effects (see Table 1). Given their cardioprotective benefits, ACEIs should therefore be the preferred agents in antihypertensive combination therapies, which include agents that inhibit the renin–angiotensin system.
Blood Pressure Control – The Key Parameters
It is well known that BP varies throughout the day and between seasons,28 and several repeated measurements are necessary over weeks/months for a reliable diagnosis of mild-to-moderate hypertension.29 BP values can be obtained at the clinic, in an ambulatory mode or by self-measurement. Several factors may interfere with measurements. Some involve the patient and include talking during measurement, exposure to cold, alcohol ingestion and the ‘white coat effect’. Other such interfering factors arise from the measurer (expectation bias) and the technique itself (patient’s position, arm position and support, cuff size).30 Out-of-office measures are likely to offer more stable and reproducible estimates of BP and correlate better with prognosis than in-office measures. In fact, ambulatory blood pressure monitoring (ABPM) is superior to in-office measurement for predicting CV events31 and is currently the gold standard in BP monitoring.32
According to the British Hypertension Society, assessment of BP and associated risk factors is an important component of CV risk and target organ damage estimation. Target organ damage evaluation involves determination of the albumin–creatinine ratio, plasma glucose, electrolytes, estimated glomerular filtration rate and cholesterol levels, as well as assessing for retinopathy and cardiac structure and function. More specifically, subclinical target organ damage can be evaluated by echocardiography (left ventricular hypertrophy), ankle–brachial index (ABI), arterial stiffening index (AIS) and pulse wave velocity (PWV) calculation and central BP measurement.
There are a large number of methods for measuring BP: central BP (aortic, carotid), brachial BP (systolic, diastolic, mean, pulse pressure), 24-hour BP (ABPM, home BP monitoring) and BP variability (24-hour, within-visit, visit-to-visit). However, current guidelines for hypertension management disagree over the use of these methods for BP assessment. When analysing the validity of a BP measurement method and resultant parameter, it is important to take into account the significance for prognosis, the possibility of improvement and the difference between antihypertensive drugs.
Brachial BP reduction correlates with favourable CV outcomes (stroke, CV death, CHD and all-cause mortality).33 According to data from ASCOT,12 reductions in SBP can only explain half of the reduction in CV. Similar differences in brachial SBP in different trials can result in different clinical outcomes. For example, in ASCOT, the total mortality reduction was 11 % and the CV mortality reduction was 24 % for a 2.7 mmHg SBP difference. However, there were no changes in the mortality rates for SBP differences of 2.4, 4.0, 2.0, 2.0 and 3.3 mmHg in the ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET),34 Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease (TRANSCEND),35 Prevention Regimen For Effectively avoiding Second Strokes (PRoFESS),36 Valsartan Antihypertensive Long-Term Use Evaluation (VALUE)37 and Randomized Olmesartan and Diabetes Microalbuminuria Prevention (ROADMAP)38 studies, respectively.
A meta-analysis of 11 longitudinal studies involving 5,648 participants followed-up for 45 months showed that central BP is a powerful predictor of CV outcomes, with relative risks of 1.094 (95 % CI 1.019–1.175) and 1.137 (CI 1.063–1.215) per 10 mmHg increase of central SBP and pulse pressure, respectively.39 In the ASCOT Conduit Artery Function Evaluation (ASCOT-CAFE), similar values of brachial SBP were obtained with a combination of amlodipine/perindopril and b-blocker/diuretic (difference=0.7 mmHg, p=0.07), but a further reduction in central SBP measurements was observed with amlodipine/perindopril (difference=4.3 mmHg, p<0.0001).40
The Dublin outcome study determined that ABPM (nocturnal) is superior in the prediction of five-year risk of CV death compared with daytime BP, 24-hour BP and conventional office BP.41 Relevantly, amlodipine combined with perindopril reduces night-time SBP compared with atenolol combined with thiazide (difference=2.2 mmHg, 95 % CI -3.4 to -0.9, p=0.0008), but not night-time DBP (difference= 0.8 mmHg, CI 0.0 to -1.6, p=0.0523).42
Finally, SBP variability correlates with mortality risk and is a strong predictor of CV (stroke and CHD) events. Data on 956 US adults ≥20 years of age from the Third National Health and Nutrition Examination Survey (NHANES III) revealed that a standard deviation higher than 8.3 mmHg is associated with a 50 % increase in mortality risk versus a deviation inferior to 4.8 mmHg.43 Moreover, residual visit-to-visit variability has a better prognostic value than mean brachial SBP (e.g. top-decile HR for stroke: 3.25, 95 % CI 2.32–4.54, p<0.0001) in ASCOT-BPLA.44 Moreover, amlodipine/perindopril showed superior and sustained mean within-visit SBP variability reductions over a five-year follow-up period compared with atenolol/bendroflumethiazide.45
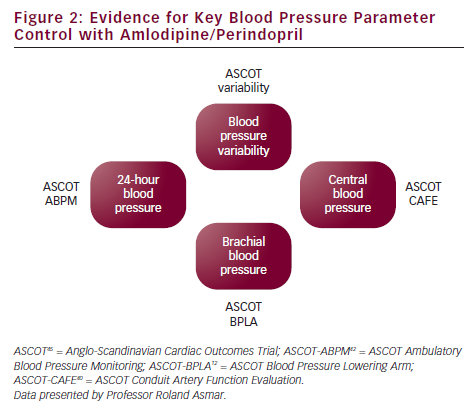
In summary, there is substantial evidence for superior control of key BP parameters with the amlodipine/perindopril combination in the various ASCOT substudies: BP variability in ASCOT,45 brachial BP in ASCOT-BPLA,12 central BP in ASCOT-CAFE40 and 24-hour BP in ASCOT-ABPM42 (see Figure 2). Remarkably, an adequate control of these specific key BP parameters with these two drugs has been shown to protect against the development of resistant hypertension, with a relative risk reduction of 43 % (HR=0.57, 95 % CI 0.50–0.60, p<0.001).46 It is therefore important to determine which BP parameter should be controlled in order to evaluate the efficacy of a therapeutic strategy and discuss prognosis and treatment options.
Other Strategic Challenges Besides Blood Pressure Lowering
An analysis of changes in prevalence and control of hypertension in the US showed a 30 % increase in the prevalence rate over a total of 10 years, from 1988 to 1994 through 1999 to 2004, despite the implementation of preventive measures. The number of patients with controlled BP improved overall in all ethnic and gender groups surveyed (26 % for the period of 1988 to 1994 to 35 % for 1999 to 2004, p<0.001), but increases in awareness were modest (69–72 %, p=0.04).47
The European Study of Cardiovascular Risk (EURIKA) revealed that approximately 35 % of hypertensive patients remain at high CV risk and 65 % remain at risk (10-year risk of CV death ≥5 % based on the Systematic Coronary Risk Evaluation), despite achieving treatment goals values comparable to conditions such as dyslipidaemia, type 2 diabetes and obesity.48 Differences in compliance according to drug class and type of therapy (i.e. monotherapy/combination therapy) can be observed and may partially explain why patients are not achieving treatment goals. Rates of treatment discontinuation are equivalent for ACEIs and ARBs, but are higher for CCBs, diuretics, antisympathetic drugs and b-blockers. Different fixed-dose combinations of ACEIs, ARBs and b-blockers with diuretics, as well as associations of distinct diuretics, also show higher discontinuation rates, ranging from 10 to 25 %.49 Additionally, compliance and persistence rates are higher for fixed-dose than free-drug combinations (overall OR=1.29, 95 % CI 1.11–1.50, p=0.08), despite achieving similar BP levels and number of adverse events (OR=1.30, CI 0.98–1.71, p=0.533; and OR=0.80, CI 0.58–1.11, p=0.584, respectively).8
The presence of co-morbidities in hypertensive patients cannot be ignored. The Brisighella and Framingham studies identified lipid disorders as a main risk factor for hypertension; other factors comprised abnormal glucose metabolism (impaired fasting glucose, impaired glucose intolerance), diabetes mellitus, metabolic syndrome, obesity, presence of target organ damage and family history. In fact, more than 50 % of patients with high BP present with two or more risk factors.50,51 Consequently, simultaneous BP and total cholesterol reductions significantly improve the prognosis of patients on antihypertensive agents in terms of CV morbidity (2 % CV event rate for a combined reduction of 20 % in SBP and 11 % in total cholesterol versus 10 % for a 1 % reduction in SBP alone, and 15 % for a 2 % reduction in cholesterol levels alone). However, BP reductions per se cannot lower the rate of events when serum cholesterol levels increase or remain unchanged (CV event rate over 25 % for an increase in cholesterol levels of 7 %, even if accompanied by a 20 % reduction in SBP).52
Not unexpectedly, administration of a statin (atorvastatin) as an add-on to an antihypertensive regimen showed superior BP and sustained CV events reductions. As found in the ASCOT-Lipid Lowering Arm (LLA), there was a 36 % risk reduction in non-fatal MI and fatal CHD (HR=0.64, 95 % CI 0.50–0.83, p=0.0005) and a 27 % decrease in fatal and non-fatal stroke (HR=0.73, CI 0.56–0.96, p=0.0236).53 Notwithstanding, these beneficial effects strongly differed between antihypertensive combinations. Atorvastatin reduced the relative risk of CHD events by 53 % (HR=0.47, CI 0.32–0.69, p<0.0001) among those randomised to amlodipine/perindopril versus 16 % (HR=0.84, CI 0.60–1.17, p=0.30) among those taking atenolol/thiazide (overall p-value of 0.025), but there were no significant differences in total cardiovascular events or strokes for the amlodipine- (HR=0.73, CI:0.60–0.88, p=0.001; and HR=0.69, CI 0.45–1.06, p=0.09, respectively) and the atenolol-based regimens (HR=0.85, CI 0.71–1.02, p=0.08; and HR=0.76, CI 0.53–1.08, p=0.01, respectively).54
This synergistic effect between BP- and lipid-lowering regimens may be due to transcriptional and post-transcriptional regulation of the AT1R gene by insulin and low-density lipoprotein (LDL), which upregulate the expression of the receptor by increasing transcript stability in vitro.55 It was shown recently that the vascular-protective properties of high-density lipoprotein (HDL) are partially explained by its inhibitory effect on diabetes-induced AT1R levels and the subsequent decrease in reactive oxygen species that results in increased NO bioavailability and improved endothelial function.56
In a similar way to the observed additive benefits of double targeting cholesterol and BP, data from the ADVANCE study show that total and CV mortality is further reduced in patients with both hypertension and diabetes when BP and glycaemia are controlled at the same time (RRR=18 %, p=0.04; and RRR=24 %, p=0.04, respectively).57 Nonetheless, not all hypertension treatments are alike in their protective effects: amlodipine/perindopril seems to perform better than atenolol/thiazide in terms of new onset of diabetes (HR=0.70, 95 % CI 0.63–0.78, p<0.0001), as reported in ASCOT.12
Even within the same class, antihypertensive drugs can have distinct pharmacological properties, which may be responsible for the variability observed in activity. Differences in the potency of ACEIs can be attributed to their pharmacokinetic and pharmacodynamic profiles. For example, plasma ACE inhibition is almost non-existent 48 hours post-administration of enalapril or cilazapril, when compared with a 40 % inhibition rate for perindopril.58 In addition, a differential affinity for tissues can be observed in this drug class, with higher relative tissue potency for heart homogenates shown by active metabolites perindoprilat, quinaprilat and ramiprilat.59
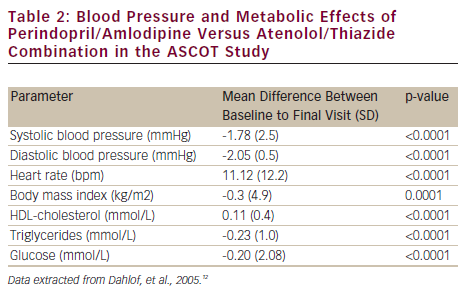
These exceptional properties of perindopril may result in the superior outcomes observed for combinations including specifically this ACEI. As shown in ASCOT, the amlodipine/perindopril combination not only reduces SBP and DBP (-1.78 and -2.05 mmHg, respectively) but also reduces all-cause and CV mortality significantly during a follow-up period of five years versus atenolol/thiazide (738 versus 820 events, p=0.025; and 263 versus 342 events, p=0.001, respectively). Moreover, this long-acting combination effects significant changes in HDL-cholesterol, triglycerides and glucose levels (mean differences of 0.11, -0.23 and -0.20 mmol/L, respectively) (see Table 2).12
The long-term beneficial effects of multifactorial intervention on mortality and CV events are obvious in the Steno-2 study. The absolute risk reduction of CV events was 29 % (HR=0.41, 95 % CI 0.25–0.67, p<0.001) after 13 years of intensive therapy, involving BP, glucose, lipid and plaque formation control, compared with conventional therapy, and the absolute reduction in risk of mortality was 20 % (HR=0.54, CI 0.32–0.89, p=0.02).60 Thus, an effective BP control must be accompanied by strict management of concomitant risk factors, and this can be successfully achieved with combination strategies that affect patients' BP parameters and metabolic profiles.
Conclusion
Hypertension is associated with an increased risk of CV disease regardless of age, gender or ethnicity, and control of BP can significantly lower this risk. Unfortunately, not all patients achieve optimal values on monotherapy. Current treatment guidelines therefore recommend a step-by-step care approach where combination therapy has an increasingly predominant role.
Adequate BP control can only be achieved in most patients by using complementary antihypertensive agents. The choice of type and dose of agents determines the efficacy, safety and compliance of the combination, and ACEIs show significant benefits in terms of mortality over ARBs. Combination therapy also allows adequate BP control across all subgroups of patients, including those unresponsive or intolerant to specific antihypertensive agents.
Combination therapy is thus an effective treatment strategy for the control of hypertension in newly diagnosed or advanced patients. Independent of the suitability of each patient-specific combination, treatment goals should always be kept in mind, not only to control BP measurements but also to prevent CV events and reduce CV risk and mortality.