Left main coronary artery disease (LMCAD) is identified in about 5% of patients undergoing coronary arteriography for the assessment of obstructive coronary artery disease (CAD) and is associated with increased morbidity and mortality.1 For decades, coronary artery bypass grafting (CABG) has been the gold standard treatment for LMCAD. However, with continuing advances in interventional cardiology, percutaneous coronary intervention (PCI) has become a safe and effective management option in selected patients with LMCAD.2
The XIENCE versus Coronary Artery Bypass Surgery for Effectiveness of Left Main Revascularization (EXCEL) trial assessed the safety and efficacy of CABG versus PCI for the management of LMCAD. The authors reported their 3-year follow-up data in December 2016 and 5-year follow-up data in September 2019.3,4
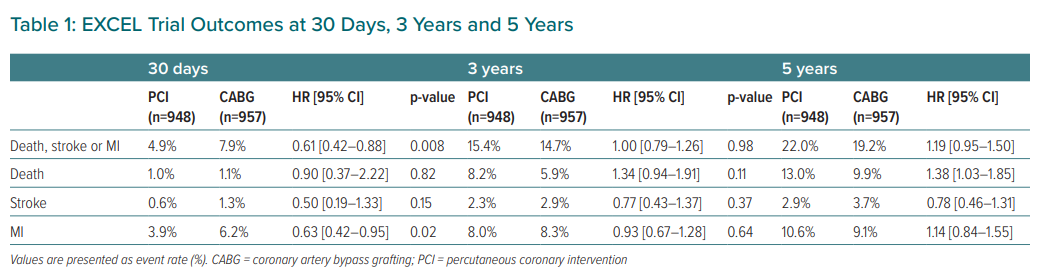
In this contemporary, non-inferiority trial, 1,905 patients with low or intermediate anatomical complexity LMCAD were randomly assigned to undergo either PCI with fluoropolymer-based cobalt-chromium everolimus-eluting stents (PCI group, n=948) or CABG (CABG group, n=957). Results at 30-day, 3- and 5-year follow-up are shown in Table 1. The authors concluded that treatment with PCI and CABG did not differ significantly regarding the composite outcome of death, stroke or MI in patients with LMCAD of low or intermediate anatomical complexity.
Although the EXCEL trial has many strengths including its recruitment and successful follow-up of a large number of patients, the interpretation of its findings has generated controversy and the lead surgical investigator withdrew his name from its final publication. Moreover, in December 2019, the European Association for Cardio-thoracic Surgery council withdrew its support for the treatment recommendations on LMCAD published in the 2018 European Society of Cardiology/European Association for Cardio-thoracic Surgery myocardial revascularisation guidelines that were largely based on the EXCEL trial findings.5
A number of issues in the trial have been extremely controversial and debated by the scientific community worldwide. Two main issues have captured the attention of clinicians, researchers and patients alike:
- The reporting of the overall results as ‘neutral’ at 5-years follow-up with similar outcomes for CABG and PCI treatments.
- Discrepancies that emerged after the publication of the study regarding the definition of periprocedural MI used in the trial.
‘Neutral’ Overall Results
The authors reported that in patients with LMCAD and a low and intermediate SYNTAX score, the study’s composite primary endpoint – death, stroke or MI – at 5 years was 22% in the PCI group and 19.2% in the CABG group (p=0.13) and concluded that there were no significant differences between revascularisation with PCI or CABG. Moreover, all-cause mortality was reported as being essentially equal between the PCI and CABG groups at 1 and 2 years but it began to diverge in a sustained fashion such that endpoint rates became 13.0% versus 9.9% (OR 1.38; 95% CI [1.03– 1.85]) by 5 years. The mortality rate at 5 years was 38% higher in the PCI arm compared to the CABG arm.
To provide additional insights into the interpretation of data reported by EXCEL, Bayesian methods were proposed, which give probability estimates of clinical interest and allow the consideration of pre-existing evidence and how it can influence test results and aid medical decision-making.6 These methods have been recommended to assist in the interpretation of clinical trials for more than 20 years.7 Reanalysis of the EXCEL trial data with the use of Bayesian methods resulted in the opposite conclusions to what was initially reported by the EXCEL investigators: the mean difference regarding incidence of the primary composite outcome was 3% lower in the CABG group compared to the PCI, whereas mortality was 1% lower with CABG than with PCI at 5 years. Similar results were reported in a systematic review of all previous CABG versus PCI studies. This analysis suggests that long-term results with the use of PCI are inferior to CABG in patients with LMCAD.8
Discrepancies in the Different Definition of MI in the EXCEL Trial
In the EXCEL protocol, the universal definition of MI from the joint European Society of Cardiology/American College of Cardiology/American Heart Association/World Heart Federation Task Force for the Redefinition of MI was a pre-specified secondary endpoint. However, the EXCEL investigators used the Society for Cardiovascular Angiography and Interventions (SCAI) definition of MI when the results were reported. This is problematic, as the universal definition was a pre-specified secondary endpoint in the original EXCEL protocol and this definition was not used in the 3-year or 5-year publications.
Post-revascularisation myonecrosis was assessed after PCI and CABG by serial measurements of creatine kinase-MB (CK-MB) and defined using an identical threshold for PCI and CABG (CK-MB elevation >10 × the upper reference limit (URL) within 72 hours post-procedure, or >5× URL with new Q waves, angiographic vessel occlusion or loss of myocardium on imaging). Troponin assessments were optional and, according to the authors, were collected in a minority of patients, which seriously compromises the diagnosis of procedural MI using alternative definitions.
In a letter to the editor published in the New England Journal of Medicine in July 2020, the authors provided evidence on the cumulative incidence of MI at the 5-year follow-up using the third universal definition of MI and a large discrepancy has become apparent in the number of CABG-related procedural MIs when data using these two different definitions are compared.9,10 Unfortunately, the letter did not include a calculation of the primary composite outcome using the universal definition of MI.
Conclusion
The evidence indicates that the jury is still out as to whether CABG or PCI is a better treatment option for patients with LMCAD and the issue continues to be debated. A review process initiated by the NEJM is under way and it is hoped that its conclusions will help gain insight into the true findings of the EXCEL trial.