Intensive basic and clinical research over the past 50 years resulted in a progressively better understanding of ischaemic heart disease, its diagnosis and treatment, with a sustained reduction in mortality in both acute coronary syndromes and during the chronic phase of the disease.1,2 However, better long-term prognosis and aging of the population are responsible for a continuous worldwide increase in the prevalence of cardiovascular disease, particularly chronic ischaemic heart disease, with stable angina being the most frequent complaint in clinical practice.3
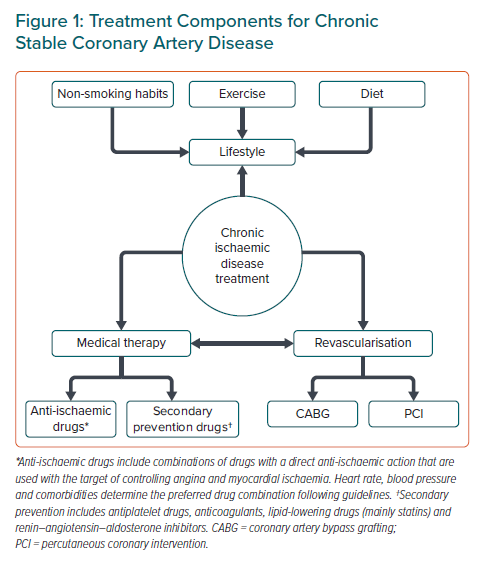
Modern treatment of myocardial ischaemia is based on three main pillars: lifestyle, medical therapy and myocardial revascularisation (Figure 1). All are equally important and none of them can be ignored. The quick evolution of knowledge in the field underlines the paramount importance of guidelines and patient education.
The benefit of prompt myocardial revascularisation has been well established in acute coronary syndromes, particularly in patients presenting with chest pain and ST-elevation MI (STEMI). However, despite the ever-increasing number of revascularisation procedures, the potential benefit of coronary angiography and revascularisation in chronic stable patients remain controversial.4–6 Thus, the International Study of Comparative Health Effectiveness with Medical and Invasive Approaches (ISCHEMIA) trial was designed to determine the effect of adding cardiac catheterisation (hereafter ‘angiography’) and revascularisation, when feasible, to medical therapy in patients with stable coronary disease and moderate or severe ischaemia.7
What is the ISCHEMIA Trial?
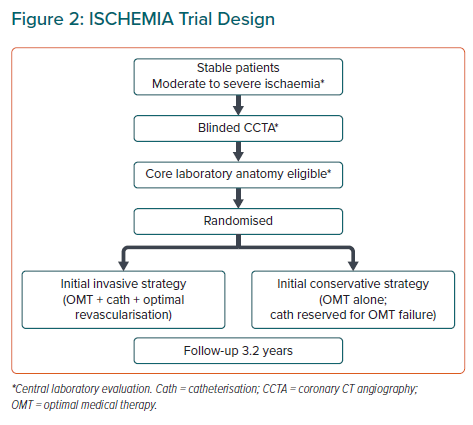
The ISCHEMIA trial was a prospective multicentre randomised study supported by the US National Heart, Lung, and Blood Institute. The study compared two initial treatment strategies in chronic stable patients with moderate to severe myocardial ischaemia identified during stress testing, namely an initially invasive strategy (INV) with cardiac catheterisation and revascularisation in addition to optimal medical therapy (OMT) and an initially conservative strategy (CON; i.e. OMT), with coronary angiography and revascularisation reserved for patients with angina incompatible with normal life or acute coronary syndrome episodes.7 The main objective of the ISCHEMIA trial was to demonstrate that initial INV was superior to CON in improving relevant clinical outcomes, including cardiovascular death, MI, hospital admission for unstable angina, heart failure or resuscitated cardiac arrest (Figure 2). The design of the ISCHEMIA trial followed the contemporary standards used in multicentre clinical trials.7
Patients with moderate to severe myocardial ischaemia (>10% of the left ventricle) were selected for screening. Blinded coronary CT angiography (CCTA) identified patients with significant coronary stenosis in at least one epicardial vessel as candidates for the trial, excluding cases with non-protected left main stem stenosis (>50%) and patients without significant stenosis in the epicardial coronary arteries.7 Patients with a recent episode of acute coronary syndromes, New York Heart Association Class III–IV heart failure, left ventricular ejection fraction (LVEF) <35%, unacceptable angina according to patients preferences to perform a desired life activity and previously determined non-revascularisable coronary anatomy were also excluded.7 Patients with advanced kidney disease (estimated glomerular filtration rate <30 ml/min/1.73 m2 or on dialysis) were included in a parallel randomised trial (ISCHEMIA-CKD) that had the same design except that no CCTA was use to select eligible cases.8
Patients were randomly assigned to INV or CON (Figure 2). The primary outcome of the ISCHEMIA trial was a composite outcome of cardiovascular death, MI, hospitalisation for unstable angina, heart failure or resuscitated cardiac arrest. The mean follow-up period was 3.2 years.7
Participating hospitals were required to demonstrate experience with both percutaneous and revascularisation procedures. The use of pressure guidewires and fractional flow reserve (FFR) measurement in borderline lesions, third-generation drug-eluting stents (DES) and complete revascularisation were strongly recommended. Decisions regarding surgical or percutaneous revascularisation were made for each patient individually according to coronary anatomy and heart team discussion. Frequent quality controls benchmarked the use of medications and target levels of blood pressure, non-smoking status, LDL and glycaemic levels according to guideline recommendations.7
Was the ISCHEMIA Trial Needed?
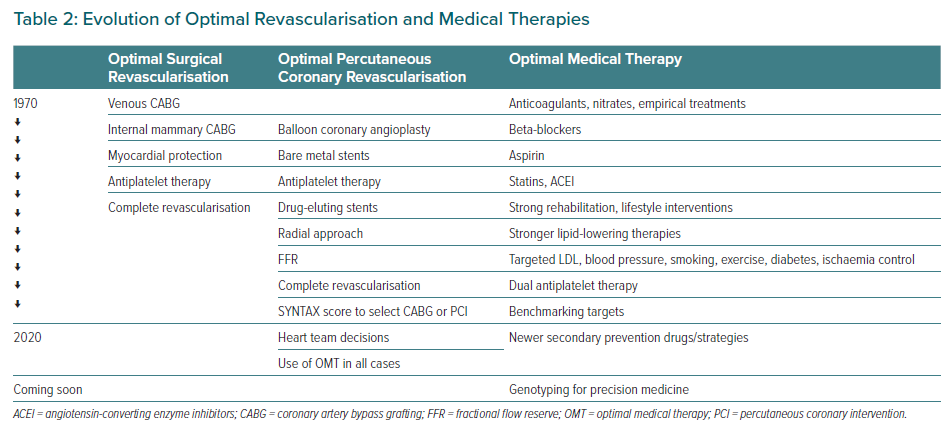
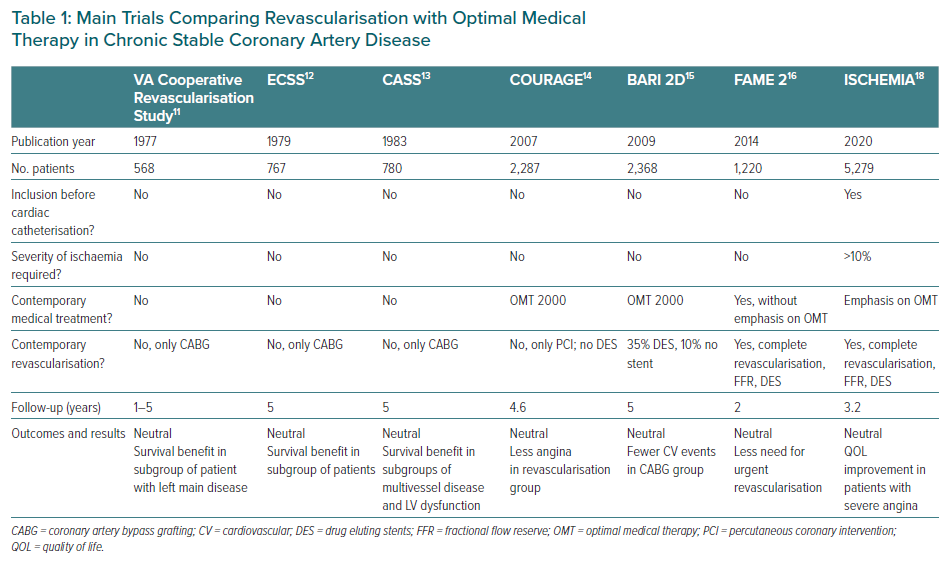
The clear answer is yes. Clinical guidelines recommend revascularisation in stable high-risk patients, particularly if the myocardium at risk is >10%.9,10 These recommendations are based primarily on evidence from classic clinical trials, including Veterans Administration Cooperative Revascularisation Study, ECSS and CASS, with less evidence of benefit in more recent trials, such as COURAGE, BARI 2D and FAME 2.11–16 In all trials, revascularisation improved angina symptoms, but better clinical outcomes were never demonstrated with the exception of the early trials, particularly in the presence of heart failure or if the myocardium at risk was >10% and a reduced need for revascularisation procedures during follow-up in the FAME 2 trial (Table 1).9,14 In addition, an ischaemia severity threshold was not defined, a fact that may have led to the selection of patients in whom revascularisation was unlikely to be of benefit.17 In addition, in all previous trials, patients were selected according to coronary artery anatomy, with the opportunity of selecting the best suitable cases for revascularisation and thus imposing a bias to generalise recommendations in broad populations. Table 2 summarises the evolution of optimal revascularisation and medical treatment, including the contemporary state of the art, still in constant evolution. Certainly, there has been considerable progress in medical treatments and interventions, somehow making the results of trials conducted many years ago obsolete.
For these reasons, a trial comparing an invasive to a conservative strategy was clearly needed. The ISCHEMIA trial avoided some of the design deficiencies of previous trials and represents the largest trial using contemporary revascularisation procedures and OMT to test the hypothesis that early initial INV plus OMT is better than OMT alone.
Neutral Results
The median age of participants in the ISCHEMIA trial was 64 years; 41% had diabetes, 19% had prior MI and 90% had prior angina.18 Stress imaging was the qualifying test for 75% of participants (86% with moderate or severe ischaemia); of those in whom CCTA was performed, 79% had multivessel disease, 87% had left anterior descending coronary artery disease and 47% had proximal left anterior descending coronary artery disease. There were no differences between the INV and CON groups.18 Therefore, although the study population could not be considered as low risk, certainly the highest-risk population (low LVEF, unstable angina and unprotected left main disease) was excluded.
The results of the ISCHEMIA trial, in which participants were followed for a median of 3.2 years, summarised in Table 3.19 There were no differences between the INV and CON groups for the primary endpoint (a composite of death from cardiovascular causes, MI or hospitalisation for unstable angina, heart failure or resuscitated cardiac arrest), and similar results were found in the ISCHEMIA-CKD trial.8 The rate of hospitalisations for unstable angina was lower, but that for heart failure was higher, in the INV group, but the differences did not reach statistical significance.19 Subgroup analysis also failed to demonstrate differences between INV and CON in patients with prior MI or previous revascularisation or according to LVEF, the severity of myocardial ischaemia or coronary artery anatomy, adding consistency to the main results of the trial.19 Overall, the superiority of an initial INV could not be demonstrated.
However, there was a benefit favouring INV: an improvement in angina control and quality of life metrics in patients who had severe angina (daily/weekly), with no improvement in patients with less frequent or no angina.20
What Are Optimal Revascularisation and OMT?
Standards for both revascularisation procedures, anti-ischaemic therapy and secondary prevention strategies have evolved considerably (Table 2).
Contemporary optimal medical revascularisation includes the selection of surgical revascularisation or percutaneous coronary intervention (PCI) according to complexity scores (the SYNTAX score being the most commonly recommended) and heart team decisions, large use internal mammary artery, the use of FFR for better evaluation of epicardial stenosis, complete revascularisation of significant coronary artery stenosis, the use of radial artery and DES during PCI and, most importantly, concomitant OMT in all cases.10
Today, OMT includes strong lifestyle programs for non-smoking, exercise and healthy diet; the routine use of antiplatelet agents and lipid-lowering drugs (mainly statins), as well as target-oriented therapies to control myocardial ischaemia using a combination of anti-ischaemic drugs; controlling LDL to levels that were unthinkable only a few years ago; controlling blood pressure; and the use of new therapies, such as newer antidiabetic agents.9,10,21,22 This approach was simply non-existent even a few years ago. Continuous improvements should be added to OMT, and older therapies discarded when they become obsolete.
The ISCHEMIA trial recommended both optimal revascularisation and OMT, which was somehow achieved in a significantly higher proportion of patient than in previous trials.
In the INV group, coronary angiography was performed in 96% of patients and revascularisation was performed in 70% (74% PCI, 24% coronary artery bypass grafting).19 No revascularisation was justified by the presence of no significant stenosis using FFR or was considered not technically feasible. Surgery was selected in 26% of patients, with 93% undergoing arterial grafting and DES being used in 98% of PCI procedures. During follow-up, 5,337 and 1,706 revascularisation procedures were performed in the INV and CON groups, respectively. One in four patients in the CON group needed revascularisation during the follow-up period.19 These findings are similar to those reported in the more recent previous trials in which patients were selected according to angiographic coronary anatomy.
OMT was monitored at each visit, with bench marking between countries and sites participating in the trial. The control of risk factors and the use of secondary prevention medication increased throughout the follow-up period to levels higher than in contemporary registries. Furthermore, 90% of patients stopped smoking, blood pressure was controlled in 77% of patients and LDL <1.81 mmol/l was achieved in 59% of patients. High levels of optimisation of medical therapy (defined as meeting all the following criteria: LDL <1.81 mmol/l, the use of antiplatelet drugs, no smoking, systolic blood pressure <140 mmHg) increased from 20% at baseline to 40% through the trial.7,23 Of note, the level of OMT achieved in the INV and CON groups was similar.
Other Findings of Clinical Interest
Low Event Rates
Cardiovascular and all-cause deaths were very low (<2%/year).7 A low number of clinically relevant events has been reported in most contemporary trials in stable chronic heart disease patients.24,25 This may be related to extensive exclusion criteria, in particular heart failure and major comorbidities, but optimisation of contemporary medical therapy following secondary prevention guideline recommendations for the control of risk factors, rehabilitation, anti-ischaemic therapy and the routine use of drugs with evidence-based benefits, as in the ISCHEMIA trial, may have had a large impact on outcomes.
The need for revascularisation during long-term follow-up has been the leading event in some secondary prevention trials. However, it is difficult to standardise indications for revascularisation in multicentre trials, and revascularisation was deliberately not included as a primary endpoint in the ISCHEMIA trial. In the FAME 2 trial, recurrent events were not detailed, but excluding revascularisation, death or MI at 2 years, the rate of recurrence was 7.3%, without differences between groups, and the rate of death from cardiac causes was 0.7%.16,26
High Proportion of Normal Coronary Arteries
Screening of patients with moderate to severe myocardial ischaemia revealed a high proportion of patients with normal epicardial coronary arteries (21%).18 This population, recently defined as ischaemia with normal coronary arteries (INOCA), remains a challenge to our understanding the mechanisms responsible for myocardial ischaemia, and it is essential that diagnostic and treatment strategies are defined for this population.27
Coronary CT Angiography
In total, 7% of screened cases showed a significant (>50%) left main stem stenosis, and these patients were still considered a priority for myocardial revascularisation in contemporary guidelines. Non-invasive coronary angiography using CCTA was proved to be effective and may become a good strategy for the selection of patients for invasive coronary angiography and revascularisation in the presence of moderate to severe myocardial ischaemia.
Trial Limitations
Due to the nature of the study, the ISCHEMIA trial was not blinded, although the investigators were not aware of the coronary anatomy in the CCTA. The planned number of subjects to be enrolled in the study was decreased from 8,000 to 5,000 (8,518 patients were screened, but only 5,179 were randomised), and the initial composite endpoint was extended due to low recruitment throughout the study, <14% of patients having less than moderate to severe myocardial ischaemia, event rates that were lower than expected and follow-up that was shorter than originally planned.28 Nevertheless, this has been the largest trial conducted so far, and there was a consistency of results in predefined subgroups. Other strengths of the trial were that patients were selected according to an ischaemia threshold and that state-of-the-art contemporary revascularisation procedures and OMT were used.
The neutral results regarding clinical outcomes should be interpreted in the context of quality of life, with a reduction in angina in the subgroup of patients with more frequent and severe symptoms.
Change in Guidelines?
Current European guidelines for myocardial revascularisation recommend revascularisation on top of medical therapy in stable patients with large areas of ischaemia (≥10%) in the left ventricle or if patients have a high-risk clinical profile (Class IB).9,10 The American Heart Association guidelines also recommend revascularisation in patients with severe ischaemia according to clinical characteristics and stress testing.29 In clinical practice, the indications for cardiac catheterisation go beyond these recommendations: revascularisation is performed in some patients without evaluating the severity of the myocardial ischaemia, as well as in patients with mild or no angina. The results of the large multicentre ISCHEMIA trial challenging current clinical practice cannot be ignored, and new guidelines will most probably modify the recommendation for revascularisation in stable ischaemic heart disease.
Future Research Needed to Complement the ISCHEMIA Trial
The large ISCHEMIA database will provide additional data to better understand the results and refine practical conclusions. The limitations of the trial support new research in the clinical setting of stable coronary artery disease, including investigations of the following.
The potential benefit of early INV over the long term; this is currently being explored in the ongoing ISCHEMIA-EXTEND trial (NCT04894877).
The constant evolution of OMT will improve the medical management of stable coronary artery disease. Of particular interest is the concept of precision medicine, with the identification of patients more likely to benefit with less secondary effect from a particular therapy. The main challenge will be adherence to long-term medical therapies recommended in guidelines by patients, physicians and healthcare providers.
Stable coronary artery disease in patients with heart failure remains a challenging field. A single study, the STICH trial, had initially neutral results and a significant reduction in mortality during an extended 10 years of follow-up.30,31
Left main disease is considered a clear indication for revascularisation.9,10 However, the benefit may be different in distinct clinical settings (non-protected stenosis, SYNTAX score, severity of myocardial ischaemia, collateral circulation, progression of atherosclerosis over time, comorbidities). Even if a controlled trial may not be possible, more information is needed and real-life registries and data analysis may provide relevant insights.
Myocardial ischaemia with normal epicardial coronary arteries remains a challenge, with a better understanding of its physiopathology, diagnosis and treatment still needed.27
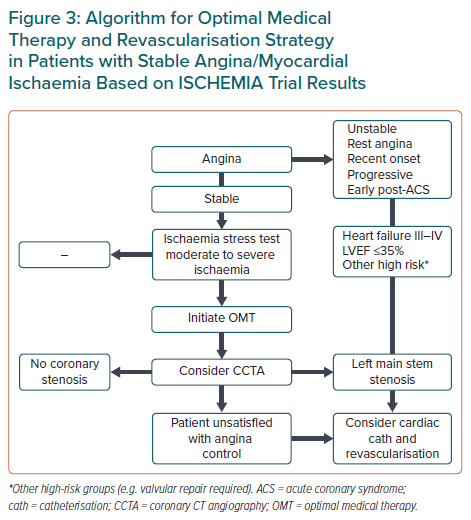
Conclusion
Revascularisation remains an important component of treatment in patients with chronic ischaemic disease. The results of the ISCHEMIA trial can only be applied to stable patients without angina or those with mild, acceptable angina during normal life. Revascularisation indications for patients excluded from the trial, namely unstable angina, unacceptable stable angina, low LVEF and episodes of acute coronary syndromes, remain unchanged.
The evaluation of a patient with chest pain should include an ischaemia test and, if mild or severe ischaemia is identified, CCTA could be the preferred strategy to exclude left main disease, as well as patients without epicardial coronary stenosis who do not require invasive cardiac catheterisation.
In stable patients, OMT is a priority and revascularisation should be considered only in patients with uncontrolled, unacceptable angina with medical treatment alone (Figure 3).