Refractory angina (RA) is conventionally defined as a chronic condition (≥3 months in duration) characterised by angina in the setting of coronary artery disease (CAD), which cannot be controlled by a combination of optimal medical therapy, angioplasty or bypass surgery, and where reversible myocardial ischaemia has been clinically established to be the cause of the symptoms.1
In clinical practice, patients diagnosed with RA are a heterogeneous group; common among them, however, is that they remain significantly limited by persistent debilitating chest discomfort despite optimised conventional therapy. In many cases, functional imaging may not demonstrate myocardial ischaemia. It is important to recognise that irrespective of aetiology, patients with refractory chest discomfort often attribute their symptoms to be cardiac in origin and believe that they may herald a life-threatening cardiac event. This predisposes to a progressive decline in their mental wellbeing and increasing anxiety whereby pain begets pain. Consequently, patients can develop persistent symptoms and pessimistic health beliefs, translating to negative behaviours and an impaired quality of life. In this regard, a shift in our approach of RA to that of managing a ‘chronic chest pain syndrome’ may help us not only to better appreciate the multifactorial aetiologies that are in operation in any given patient but also encourage the use of a holistic approach to manage these patients more effectively.
Epidemiology
Precise estimates of the prevalence and incidence of RA are not available; however, several sources suggest that this is a large and growing problem.2,3 Variations in definition and clinical heterogeneity of patients labelled with a diagnosis of RA significantly complicate such endeavours. Data from the Canadian Community Health Survey (2000–2001) suggest that ~500,000 Canadians are living with unresolved angina.4 The proportion of these patients with true RA is unknown.5 In the US, it is estimated that between 600,000 and 1.8 million patients have RA, with approximately 75,000 new cases diagnosed each year.5,6 In Europe, the annual incidence of RA is estimated at 30,000–50,000 new cases per year.1,7 Specific figures for the UK are lacking and further work to define the burden of RA in the UK population is needed. However, if the results shown by Williams et al., who found that 6.7 % of patients undergoing angiography in a contemporary cohort had no revascularisation option, are applied to the 247,363 angiograms performed in England in 2014, it can be estimated that ~16,500 new cases of RA may occur in England per year.3,8 Given improvements in CAD-related survival and increasing age of the population, together with an increasing appreciation from the contemporary Outcome of Percutaneous Coronary Intervention for Stent ThrombosIs Multicentre Study (OPTIMIST) registry that the long term prognosis of RA is not as bad as previously thought, the incidence and prevalence of RA is only set to rise.5,6,9,10 Furthermore, it has also been recently shown by Povsic et al. that patients with RA use significant healthcare resources, undergoing frequent hospitalisations, and experience high healthcare costs (~US$10,185 over a 3-year period).11
Diagnosis
Patients with RA are a heterogeneous group that remain significantly limited by persistent debilitating chest discomfort despite optimal conventional therapy.1 This syndrome characteristically involves angina-type pain usually in the context of epicardial CAD with or without demonstrable ischaemia. Given the heterogeneity in patients complaining of chronic chest pain, appropriate stratification of patients with regard to their risk of RA helps to direct healthcare resources tailored to individual patients’ needs (see Figure 1).
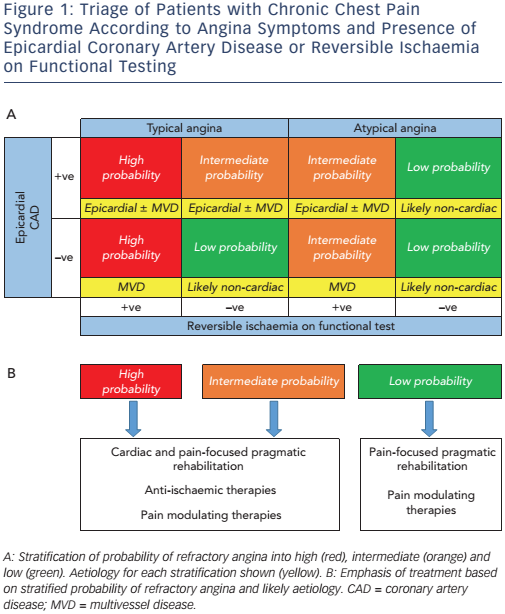
A convincing clinical history of angina, together with circumstantial evidence supporting a diagnosis, should raise clinical suspicion. The coronary anatomy in these patients is highly variable with many having had prior revascularisation (72.4 % by some estimates), and myocardial ischaemia is often difficult to detect using conventional stress imaging protocols.10 The absence of demonstrable myocardial ischaemia in the context of epicardial CAD is not uncommon in patients referred with suspected RA and should in itself not exclude the diagnosis. Interpretation of negative functional tests must therefore consider the caveat of 1) a false negative test result or 2) the degree of ischaemia lies below the detection threshold of the imaging modality employed. Furthermore, it is important to rule out a diagnosis of non-cardiac chest pain with bystander CAD. However, when a patient’s history is suggestive of angina in the absence of any other causative factor (anaemia, dyspepsia, musculoskeletal pain), the lack of demonstrable myocardial ischaemia should not absolutely preclude a diagnosis of RA.
Pharmacological Therapies
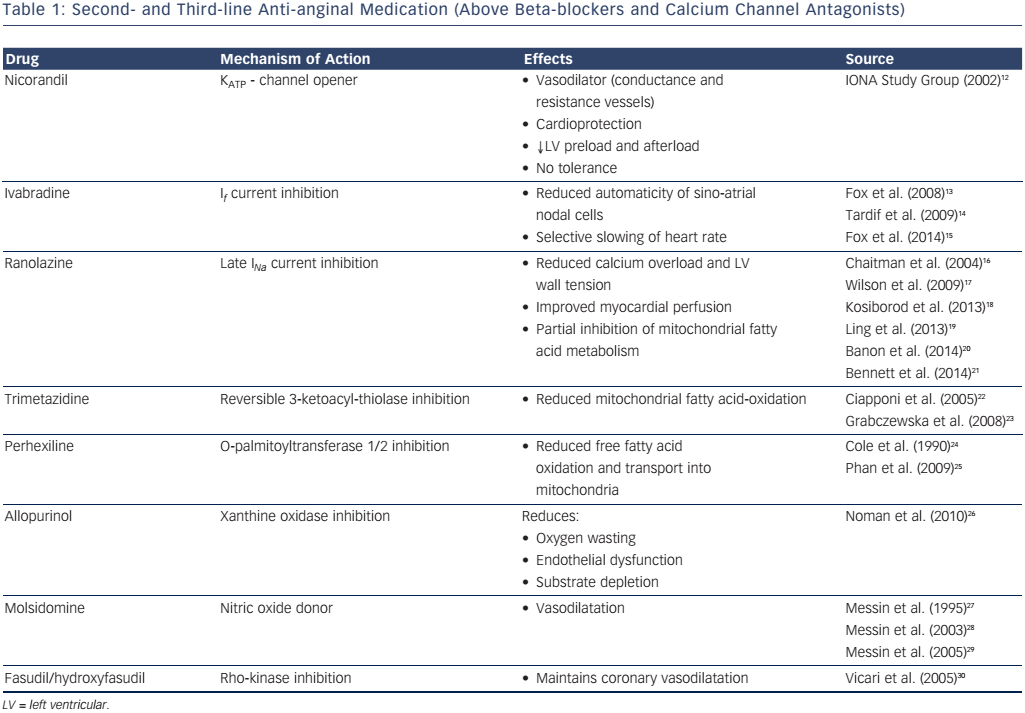
To date, no pharmacological therapy has been shown in adequatelypowered placebo-controlled randomised clinical trials (RCTs) to significantly improve symptoms and quality of life in patients with RA. However, a significant body of evidence exists in the literature with regard to pharmacological therapy for stable angina (see Table 1).12–30 The choice of additional medication over and above first-line treatment (with either a beta-blocker or calcium channel antagonist) follows the rationale for chronic stable angina – i.e. it is considered on an individual patient basis, taking into account age, heart rate, blood pressure, the presence of diabetes mellitus or impaired renal function and tolerability.31,32 Follow-up 2–4 weeks after initiation of a new medication should assess its efficacy and tolerability, and dependent on whether there is any benefit, uptitration to the maximal tolerated dose should occur. If still ineffective, the medication should be stopped and an alternative considered. However, polypharmacy is a significant problem in patients with RA and rationalising medical therapy to ensure optimal benefit, adherence and tolerability is a major clinical challenge.1,33
Coronary Sinus Reducer
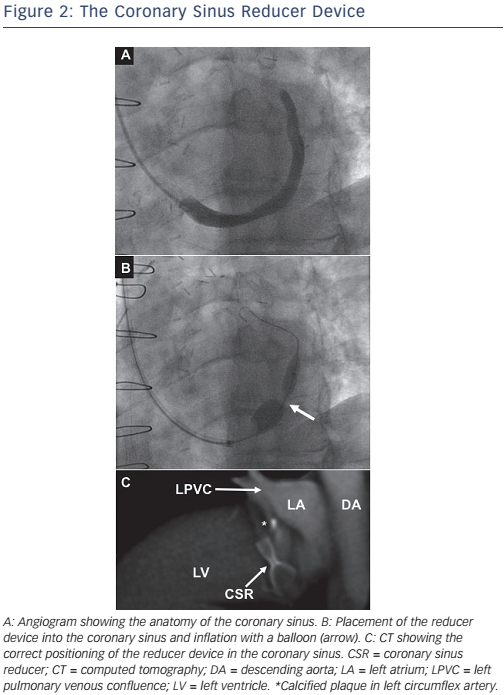
Preclinical studies have suggested that occlusion of the coronary sinus, the major venous drainage of the left heart, results in preservation of the endocardial to epicardial perfusion ratio and reduction of myocardial infarction size during coronary artery ligation.34 These data coupled with encouraging early surgical experience motivated the development of a balloon-inflatable coronary sinus reducer device, which can be implanted via a simple trans-jugular approach (see Figure 2).35–37
In normal physiology, exercise induces sympathetic vasoconstriction in the epicardial circulation, which promotes subendocardial perfusion (subendocardial:subepicardial perfusion ratio: ~1.2).38 In the setting of epicardial CAD, this mechanism is thought to become dysfunctional. Myocardial ischaemia induces impaired regional wall motion and increased left ventricular end-diastolic pressure causes compression of the subendocardial capillaries, reducing perfusion (subendocardial:subepicardial perfusion ratio: <0.8). Following insertion of a reducer, fibrosis occurs around the waist of the hourglass-shaped stent over a period of ~6 weeks, gradually increasing coronary sinus pressure and venous backflow. This results in dilation of venules and capillary recruitment with a reduction of resistance to subendocardial flow, which promotes recruitment of collateral flow from the subepicardium into the ischaemic subendocardium. It has also been suggested that this approach may encourage neovascularisation.37
A randomised, blinded, sham-controlled trial (Coronary Sinus Reducer for Treatment of Refractory Angina [COSIRA]) assessing this device has recently demonstrated significant improvements in angina symptoms and quality of life scores.39 In the treatment group, 35 % of patients had a reduction of ≥2 CCS classes compared to 15 % in the control group (p=0.020). Quality of life, as measured by the Seattle Angina Questionnaire, improved by 17.6 (treatment) versus 7.6 points (control; p=0.030). No major adverse effects were associated with this intervention.37,39–41 However, it is only suitable for patients with leftsided coronary ischaemia. Consideration whether further intervention of the coronary sinus (e.g. cardiac resynchronisation therapy) may be indicated in the near future should be made, although notably, the waist of the reducer device can be dilated to allow such procedures.42 Further results of ongoing studies to evaluate this device are eagerly awaited.43,44
Cell Therapy
Cellular therapies have gained major interest as potential novel treatments for RA. Eight RCTs have evaluated unselected bone marrow mononuclear cells (BMCs), adipose-derived regenerative cells, CD34+ (with recently published 2-year follow-up) and CD133+ progenitor cells in patients with RA.45–54 After delivery, cellular products poorly engraft and are thought to improve ischaemia by promoting neovascularisation through paracrine effects although the precise mechanisms have not been fully elucidated.55–57
Fischer et al. performed a meta-analysis demonstrating that in patients with ischaemic heart disease unsuitable for revascularisation, BMCs significantly improve Canadian Cardiovascular Society (CCS) class (mean difference −0.55; 95 % confidence interval [CI][−1.00 to −0.10], p<0.020) and reduce frequency of weekly angina episodes (mean difference −5.21; 95 % CI [−7.35 to −3.07]; p<0.00001).58 A more recent meta-analysis by Khan et al. has confirmed these findings, further reporting that cellular therapies significantly improved a composite endpoint of major adverse cardiac events (myocardial infarction, cardiac-related hospitalisation and death) (OR 0.49, 95 % CI [0.25– 0.98]) as well as angina episodes, use of anti-anginal medication, CCS class, exercise tolerance, myocardial perfusion and arrhythmia occurrence.59 Safety has been shown to be good.59,60
While these results are certainly encouraging, such analyses collate data from small clinical trials (phase I–II) and future work should focus on adequately powered, blinded trials with use of a sham control procedure for comparison.61 A number of unresolved issues remain; namely the optimal cell type, preparation, dose and method of delivery. In addition, the effects of cell therapy may be shortlived and recent data from Mann et al. suggest a need for repeated administrations to maintain efficacy.62,63 These issues must be resolved before cellular therapy can enter routine clinical practice.
External Enhanced Counterpulsation
Alternative non-invasive therapies have been investigated for patients with angina pectoris, including external enhanced counterpulsation (EECP). This involves a series of 1–2 hour sessions over several weeks (35 hours total), during which external compressive cuffs are placed on the calves, lower and upper thighs that are sequentially inflated from distal to proximal, synchronised to early diastole, and deflated at the onset of systole.5,64 This counterpulsation effect, similar to that of an intra-aortic balloon pump, promotes retrograde aortic flow with concomitant increase in diastolic pressure, increased coronary perfusion, venous return and cardiac output. Rapid deflation of the cuffs reduces systemic vascular resistance and cardiac workload.64
Furthermore, EECP has been associated with improvement in invasive haemodynamic measures of collateral function;65,66 flow-mediated dilatation of large peripheral arteries;67,68 endothelial function;69 and mediators of inflammation and vasoconstriction.70 A number of small studies have suggested potential benefit from EECP, the largest of which is the Multicenter Study of Enhanced External Counterpulsation (MUST-EECP) trial (n=139), which showed a reduction of self-reported angina episodes by ~25 % and time to development of 1 mm ST depression increased by ~15 %.71 Importantly, quality of life was improved. A meta-analysis subsequently suggested that EECP achieved an improvement in angina by at least one CCS class in 86 % of patients with stable angina pectoris, though this analysis was not restricted to those patients with RA.72 A more recent meta-analysis has reported similar results.73 However, while this technology has recently received a Class IIa, Level of Evidence B, recommendation in the European Society of Cardiology (ESC) guidelines for the management of stable CAD, a 2009 Health Technology Assessment report and Cochrane systematic review were unable to find clear evidence of clinical or cost-effectiveness.31,74,75 In addition, this therapy has a number of contraindications, notably arrhythmias, peripheral vascular disease, aortic aneurysm and aortic stenosis.5,71 Further adequately-powered, blinded, sham-controlled RCTs specifically in patients with RA are needed.
Extracorporeal Shockwave Myocardial Revascularisation Therapy
An investigational non-invasive treatment, extracorporeal shockwave myocardial revascularisation (ESMR) therapy, involves delivering low-energy shockwaves to the border zones of ischaemic myocardium (~10 % of the high-energy counterpart used in the treatment of urolithiasis) in a series of sessions delivered over 4–9 weeks.76,77 Through inducing local vasodilatation and neovascularisation, it is thought to reduce ischaemia and improve left ventricular function.78,79 Two small RCTs in the literature have shown improvements in angina in patients with RA.80,81 More recently, a case-control study of 72 patients (43 cases) not only demonstrated safety but showed ESMR therapy to be associated with modest improvements in the stress myocardial perfusion score (p=0.002), CCS class (p=0.0002), use of glyceryl trinitrate (GTN) (p<0.030), exercise tolerance (p<0.002) and hospitalisation for angina (p<0.030).76,82 A subsequent multicentre study has also demonstrated similar results.77
Interestingly, pretreatment with ESMR therapy has been suggested to enhance the beneficial effects of BMCs delivered by intracoronary infusion in patients with ischaemic left ventricular dysfunction.83 The effects were small; however, ESMR + BMCs improved left ventricular ejection fraction by 3.2 % (95 % CI [2.0–4.4]) versus ESMR + placebo infusion 1.0 % (95 % CI [0.3–2.2]; p=0.020). Upregulation of signalling molecules (stromal cell-derived factor 1 [SDF-1] and vascular endothelial growth factor [VEGF]) by ESMR is thought to underlie this phenomenon of target tissue preconditioning that may aid progenitor cell engraftment.84 Further appropriately designed studies are needed to determine the mechanism and role of ESMR in RA.
Neuromodulation
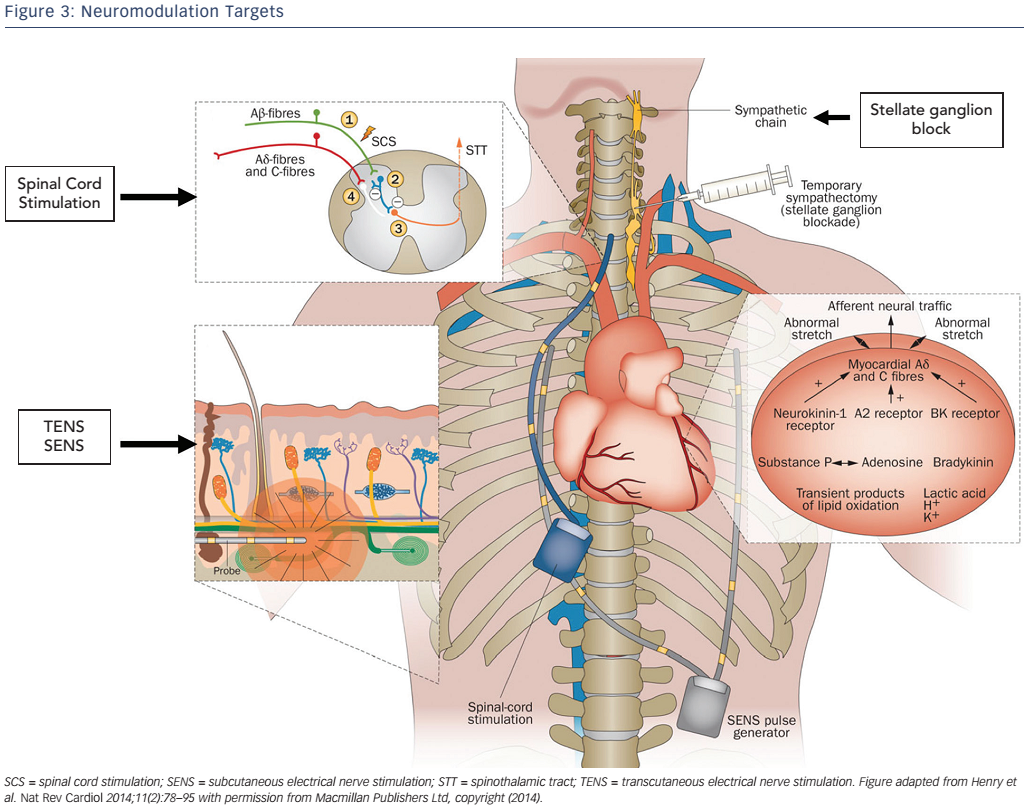
The perception of pain from visceral nociceptive stimuli is complex and the severity of symptoms is often disproportionate to the degree of ischaemia. Various approaches to modulate nociceptive signals are used in patients with RA (see Figure 3), of which, implantation of spinal cord stimulation (SCS) has received a Class IIb, Level of Evidence B, recommendation in recent ESC guidelines.6,31,85 This minimally invasive procedure involves the placement of multipolar electrodes into the epidural space to deliver an electrical current to the dorsal columns between C7 and T1.5,86,87 An implanted patientcontrolled pulse generator allows stimulation at the onset of angina, inducing paraesthesia at the location of anginal chest discomfort.
Mechanistically, SCS may result in anti-nociceptive activation of spinal afferent neurons and inhibit sympathetic efferents, attenuating vasoconstriction and reducing ischaemia.88–90 Several small scale clinical trials of SCS have been aggregated in a meta-analysis, showing significantly improved exercise capacity and quality of life with low complication rates (e.g. infection, lead displacement, etc.).91 A recent registry of 235 patients demonstrated reduced angina frequency, sublingual GTN use with SCS and improved CCS class as well as quality of life up to 1-year of follow-up.92 Adequate sham-controlled RCTs to confirm efficacy and cost-effectiveness are needed – pilot studies (e.g. Refractory Angina Spinal Cord Stimulation and Usual Care [RASCAL] study) are important but highlight the potential challenges, particularly regarding patient recruitment.93
Pragmatic Rehabilitation
Pragmatic rehabilitation is an important approach to promote patients to manage their own chest pain. Through learning cognitivebehavioural self-management techniques and challenging negative health beliefs, quality of life and psychological wellbeing can improve substantially. The Angina Plan is one such tool whereby patients’ understanding of angina can be evaluated and misconceptions corrected.94 It provides a structured approach to address maladaptive coping strategies in patients with angina and has been shown to significantly improve psychological wellbeing (anxiety and depression), symptoms (three episodes of angina fewer per week and reduced GTN use) and functional status (reduced physical limitation score and increased daily walking). Furthermore, from our experience, we have found that reassuring patients their symptoms are non-cardiac in origin has as important an impact as successful management of symptoms secondary to ischaemia.
For patients with cardiac ischaemia, pragmatic rehabilitation consists of two main components.
The first involves education to correct common misconceptions about angina and developing a basic understanding of the pain pathway. For example, the notion that stable angina in itself is not life-threatening and their pain not always cardiac in origin is emphasised. Underuse of GTN may occur due to perceptions that it may lose its effectiveness – thus such mistaken beliefs are corrected and patients encouraged to use their GTN more often.
Furthermore, it is explained that the heart can ‘adapt’ to having angina through the process of ischaemic conditioning and collateralisation.95,96 The heightened perception of death is also challenged by discussing data from the OPTIMIST Registry (n=1,200) showing that 71.6 % of patients with RA have a 9-year life expectancy.10 Additionally, patient awareness is raised about how their mental state can significantly affect their perception of symptoms.
The second component addresses important lifestyle adaptations that can significantly impact on patients’ symptoms (e.g. learning how to pace oneself, setting realistic goals and deconstructing tasks into manageable portions). Furthermore, behaviours to reduce cardiovascular risk (e.g. smoking cessation, weight loss and exercise) are strongly emphasised as is adherence to aggressive primary and secondary prevention.
Pragmatic rehabilitation is typically delivered by a nurse specialist and a clinical psychologist via a series of group-based education sessions where patients are taught self-management techniques.97 This facilitates an improvement in their quality of life whilst reducing dependency on resource-limited medical services.98 Potential avenues to develop the mode of delivery may include the use of technology such as smartphone apps. Novel technological methods to deliver such therapies have been increasingly investigated, and their benefit has been recognised for chronic conditions, including cardiovascular disease, since they can effectively complement rehabilitation as well as improve adherence to medication.99,100
The efficacy of psychoeducational interventions in patients with chronic stable angina, including RA, has been investigated in a number of small studies, which have been analysed in a meta-analysis by McGillion et al.101 Seven RCTs (total n=949) of self-management programmes were assessed, most of which studied the intervention delivered in small groups of 6–15 patients. It revealed that psychoeducational intervention resulted in significantly less angina (~3 fewer episodes per week; −2.85, 95 % CI [−4.04 to −1.66]) and reduced nitrate consumption (~4 times less per week; −3.69, 95 % CI [−5.50 to −1.89]) at 3–6 months follow-up.101 Importantly, statistically significant improvements in quality of life (as per the Seattle Angina Questionnaire) were observed for physical limitation and disease perception. More recent data from refractory angina services in the UK have reported encouraging results.97,102,103 A short psychological intervention consisting of four 2-hour sessions based on an angina programme combined with a course of cognitive behaviour therapy has been shown to significantly improve quality of life whilst reducing anxiety and depression.97 Selfreported scores of 1) restriction from and 2) control over angina also significantly improved. Impressively, these results were maintained in the long term (3-year follow-up).102
A Dedicated Multidisciplinary Service
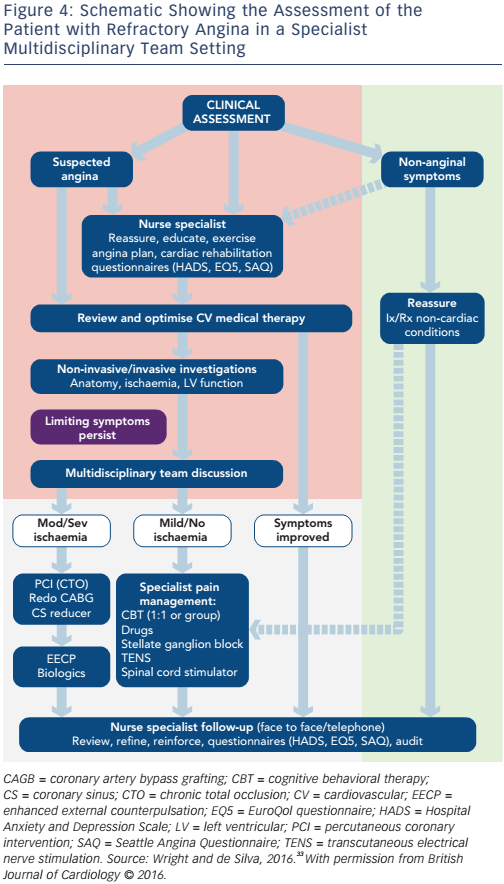
Almost by definition, the management of these ‘no option’ patients with RA is challenging. Their needs are best met via integrated care delivered by specialist multidisciplinary teams in dedicated specialist services (see Figure 4). Such a framework enables addressing the issues of this heterogeneous patient cohort in a bespoke way, and allows the full spectrum of clinical management including investigative and novel treatments for appropriately selected patients.33 Through such a combination approach involving selection from the spectrum of therapies mentioned above, management can be individually tailored to meet patients’ needs. Although such resources are scarce, the recognition of the importance of multidisciplinary teams in this unique subset of patients will hopefully encourage further provision of services.
Conclusion
Whilst novel therapeutic approaches to managing these patients are welcome, evaluation of their efficacy through robust clinical data must be rigorously pursued. The development of clinical guidelines specific to RA should also be encouraged. Finally, further studies should investigate the effect of novel therapies on reducing healthcare utilisation and demonstrate cost-effectiveness in patients with RA.